
Sometimes X-ray may interact with another orbital electron, which may be ejected from the atom. Since the process leaves a vacancy in the electron energy level from which the electron came, the outer electrons of the atom cascade down to fill the lower atomic levels, and one or more characteristic X-rays are usually emitted. The resulting daughter nuclide, if it is in an excited state, then transitions to its ground state usually gamma ray emission, but de-excitation may also take place by internal conversion. On the other hand, electron capture is always an alternative decay mode for radioactive isotopes that do have sufficient energy to decay by positron emission. 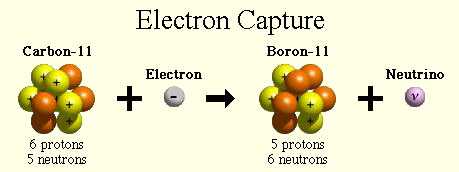
Electron capture is the primary decay mode for isotopes with insufficient energy (Q < 2 x 511 keV) difference between the isotope and its prospective daughter for the nuclide to decay by emitting a positron. This process competes with positive beta decay, which is more common for lighter nuclei. The electron is normally captured from an inner shell of an atom (K-shell). In this process a parent nucleus may capture one of its orbital electrons and emits a neutrino. In this process, a proton-rich nucleus can also reduce its nuclear charge by one unit by absorbing an atomic electron. Electron capture, known also as inverse beta decay is sometimes included as a type of beta decay, because the basic nuclear process, mediated by the weak interaction, is the same. Electron capture is a process, in which a parent nucleus captures one of its orbital electrons and emits a neutrino.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
